Most dopamine is made inside neurons, where it is quickly packed up and stored inside vesicles, tiny spherical sacks only a few millionths of a centimeter across. A protein called VMAT2 is responsible for transporting dopamine from the cytoplasm into the vesicles, where it ultimately can reach rather high concentrations. At the same time, dopamine concentrations in the cytoplasm remain low. This is probably a good thing, due to the reactive nature of the molecule.
The vesicles filled with dopamine are then ready to be used for signalling to other neurons, which involves transporting the vesicles to the correct location and causing them to fuse with the cell membrane, releasing the contents into the synaptic cleft, a small space between two neurons. After the dopamine has done its job in signaling to other neurons, it can be reused, by being taken back up into a neuron (via the dopamine transporter, DAT), which could in principle be either the same neuron where it was created or an adjacent cell. Back in the cell, it can be packaged into vesicles to be used again.
There are multiple mechanisms for disposing of dopamine, presumably to avoid accumulating an excess. Two enzymes involved in these pathways are targets of Parkinson’s drugs: monoamine oxygenase (MaoB; targeted by selegiline and rasagiline=Azilect) and COMT (targeted by entacopone=comtan=component of stalevo). That is, by reducing the rate at which dopamine is degraded, the concentration of dopamine stays higher, and Parkinson’s patients can use all the dopamine they can get.

Pathways for degrading dopamine that can be blocked by drugs. Image credit: Thank you to NEUROtiker on wikipedia!
But dopamine (and L-DOPA) is also easily chemically damaged, by oxidation, which turns it into molecules that are even more reactive, and thus dangerous, than dopamine itself. One of these is dopamine-quinone.
The fascinating mechanism for eliminating these toxic byproducts of dopamine is by forming neuromelanin, the substance that makes the substantia nigra black. Neuromelanin is a complex molecule that is quite similar to the melanin that is the primary determinant of skin and hair color.
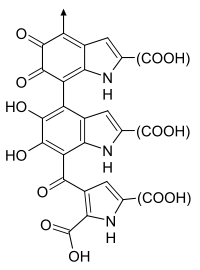
A rough idea of the chemical structure of neuromelanin. In reality, it’s a long, heterogeneous polymer.
Unlike melanin in the skin, which serves to protect the skin from sunlight, neuromelanin appears to have no useful role and instead may simply be a way of safely disposing of toxic dopamine byproducts. As such, little is present at birth, and it accumulates with age. As far as I can tell, many of the details of neuromelanin formation remain poorly understood, but the neuromelanin winds up in melanosomes, which are sort of like small membrane-bound organelles, through a process related to autophagy.

Neuromelanin. Credit: Image from this open access article.
Two interesting facts about neuromelanin and Parkinson’s:
- Parkinson’s patients have dramatically reduced levels of neuromelanin. The implications of this finding are not totally clear, but it seems consistent with neuromelanin providing protection from toxic compounds, including oxidative products of dopamine, at least as long as it stays safely encapsulated in melanosomes.
- People with Parkinson’s have substantially increased risk of malignant melanoma, despite having a decreased risk for many other cancers. Again, no one quite understands the significance of this observation, but the connections between dopamine and melanin in both the skin and brain are intriguing. [An interesting recent paper proposed a genetic link between Parkinson’s and melanoma, specifically the parkin gene, mutations in which can cause Parkinson’s, and seem to also be risk factors for melanoma; in both cases, disease is associated with decreased function of the parkin protein.]
