The information presented below has been integrated into an easy-to-use levodopa PK “calculator” at levodopalevel.com, created by Florencio Mazzoldi.
TABLE 1. L-DOPA containing drugs: There are 4 common formulations, each of which has several available dosages.
| Commonly used name (generally brand name) | “Generic” drug name(s) | Dosage form (basically what it says on the label) | Amount of L-DOPA (mg) | Fraction absorbed (Fa) |
| Sinemet | Carbidopa/ levodopa | 25-100 or 100/25 | 100 | 100% |
| Sinemet | Carbidopa/ levodopa | 25-250 or 250/25 | 250 | 100% |
| Sinemet | Carbidopa/ levodopa | 10-100 or 100/10 | 100 | 100% |
| Sinemet | Carbidopa/ levodopa | 12.5-50 or 50/12.5 | 50 | 100% |
| Sinemet CR | Carbidopa/ levodopa | 25-100 or 100/25 | 100 | 100% |
| Sinemet CR | Carbidopa/ levodopa | 50-200 or 200/50 | 200 | 100% |
| Rytary | Carbidopa/ levodopa | 23.75/95 | 95 | 70% |
| Rytary | Carbidopa/ levodopa | 36.25/145 | 145 | 70% |
| Rytary | Carbidopa/ levodopa | 48.75/195 | 195 | 70% |
| Rytary | Carbidopa/ levodopa | 61.25/245 | 245 | 70% |
| Stalevo | Carbidopa/ entacapone/ levodopa | “50″ | 50 | 100% |
| Stalevo | “ | “75″ | 75 | 100% |
| Stalevo | “ | “100″ | 100 | 100% |
| Stalevo | “ | “125″ | 125 | 100% |
| Stalevo | “ | “150″ | 150 | 100% |
| Stalevo | “ | “200″ | 200 | 100% |
Notes:
- Sinemet CR is referred to as “Continual Release” or “Extended Release”
- Regular Sinemet is sometimes referred to as “Immediate Release” to distinguish it from CR
- Rytary is a new, on-patent extended-release formulation
- Patients sometimes take half-pills of sinemet, because the low-dosage form is not commonly available. Pill splitting is less common for Stalevo, and is specifically warned against for Sinemet CR and Rytary.
References:
- http://reference.medscape.com/drug/sinemet-rytary-carbidopa-levodopa-343043
- https://www.merck.com/product/usa/pi_circulars/s/sinemet/sinemet_pi.pdf
- http://www.pdf.org/parkinson_prescription_meds
- https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/stalevo.pdf
Calculations: There are 2 key rate parameters, for absorbing the drug after ingestion (from gut to blood), and for elimination from the body. These would normally both be expressed as first-order kinetics, but examination of actual data suggests that absorption is better modeled as a zero-order process. This is a bit weird, but it is possible, for example, in the regime where the import transporter (LAT-1) is saturated, and/or the rate of absorption is limited by a zeroth-order process of the pill dissolving. I’m going to assume that the absorption time (t_abs) is the same for all doses of the same formulation, 0.5 hours for immediate release, and 1.5 hours for extended release. Rytary is more complicated, vide infra.
I will define C(t) as the time-dependent concentration of the drug in blood, with units of mg/L, D as the initial dose (mg), and t is time after the dose was taken. The relevant differential equation is dC/dt = A – kC, where A is the zeroth-order rate constant for absorption (units of mg/hour, with A=D/t_abs) and k is the first-order rate constant for elimination (units of 1/hour; k=0.693/t_half, where t_half is the half life). Note that this equation holds only until absorption is complete, at which point (t=t_abs), only elimination remains, dC/dt = -k*C. To get absolute concentrations corrects, we need to include two other factors: fraction absorbed (Fa), to account for not all of the drug getting absorbed for some formulations; and the “effective volume” of the patient, called the “volume of distribution” (Vd, estimated as 100 L for L-DOPA), in order to get concentration units.
The resulting equations are
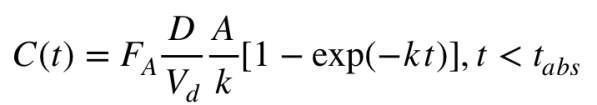
and
![]()
The relevant absorption and elimination parameters depend on the formulation and any drugs that modulate L-DOPA pharmacokinetics, as well as other factors discussed below. As shown in Table 4, the elimination half-life decreases as the disease progresses, and results will be more meaningful if patients give info about approximately their stage of disease.
TABLE 3. Absorption and elimination rate parameters for the “base case”, a typical mid-stage patient. (Early-stage patients are less likely to have problems managing their medication; many are not even on L-DOPA. As such, mid-stage patients are most likely to benefit.)
| Formulation | MaoB inhibitor (Rasagiline or selegiline) | Entacapone (from Stalevo or Comtan) | t_abs (hours) | elimination half life, t_half (hours) |
| Sinemet | – | – | 0.5 | 1.5 (base case) |
| Sinemet | + | – | 0.5 | 2.5 (base case + 1) |
| Sinemet | – | + (Comtan) | 0.5 | 2.5 (base case + 1) |
| Sinemet | + | + (Comtan) | 0.5 | 3.5 (base case + 2) |
| Sinemet CR | – | – | 1.5 | 1.5 (base case) |
| Sinemet CR | + | – | 1.5 | 2.5 (base case + 1) |
| Sinemet CR | – | + (Comtan) | 1.5 | 2.5 (base case + 1) |
| Sinemet CR | + | + (Comtan) | 1.5 | 3.5 (base case + 2) |
| Stalevo | – | + | 0.5 | 2.5 (base case + 1) |
| Stalevo | + | + | 0.5 | 3.5 (base case + 2) |
| Rytary | 0.5 (28%), 5.0 (72%) | 1.5 (base case) |
Note that the extended release formulations (Sinemet CR and Rytary) modulate the absorption rate, not elimination. The MaoB inhibitors or entacapone modulate the effective elimination half-life. Some of the numbers are guesstimates. For example, I’ve assumed that, since MaoB inhibitors and entacapone individually extend the half-life by ~1 hour each, together they extend half-life by 2 hours. It would not really make sense for patients to take Sinemet + Comtan (they would just take Stalevo instead), but this case is included for completeness.
Rytary is a little more complicated, combining immediate release and very slow release formulations in one pill. Empirically, it appears that it can be represented as the sum of 2 curves, assuming that 28% of the L-DOPA has an absorption time of 0.5 hours, and the remaining 72% an absorption time of 5.0 hours.
Below are plots of computed curves (using the ‘base case’ parameters) and corresponding experimental curves, showing fairly reasonable agreement.
To allow patients to interpret the results, shading is used to indicate the minimum therapeutic concentration and the threshold for dyskinesia. There can of course be significant patient-to-patient variability even at the same stage of disease, but the traditional Hoehn and Yahr stages are nonetheless useful:
TABLE 4.
| Disease Stage | Plain English description | Time Post-Diagnosis | Mean Therapeutic Concentration | Approximate L-DOPA effective half-life |
| Hoehn and Yahr Stage 1 | Early stage Parkinson’s with symptoms exclusively on one side | Generally within first few years after diagnosis | ~0.2 mg/L | >2 hours (4 hours might be typical) |
| H&Y Stage 2 | Symptoms on both sides, but no major problems with balance | Can remain in this stage for many years (~5-8), i.e., up to a decade post-diagnosis | ~0.4 mg/L | 1.5 hours (the base case used in Table 3) |
| H&Y Stage 3 | Onset of disabling symptoms, including impaired balance, but still able to be physically independent | Typically 1-4 years at this stage | ~0.6 mg/L | ~1 hour is typical, but can be even shorter |
| H&Y Stage 4 | Parkinson’s is severely disabling, but patient is still able to stand or walk unassisted | Typically 1-4 years at this stage | ~0.9 mg/L | Assume still ~1 hour |
| Threshold for dyskinesia (apparently roughly independent of disease stage): | [note the significant caveat that this is really only addressing peak-dose dyskinesia, which is most common] | ~0.8 mg/L |
The Wikipedia article provides formal definitions of the H&Y scale: https://en.wikipedia.org/wiki/Hoehn_and_Yahr_scale.
Other references:

